
Stepping Up and Stepping into the Clinical Trial Space
As the federal funding landscape has shifted, it’s become essential to ensure that the most promising lab studies we’ve funded have a protected pathway to become the critically needed breakthrough treatments for kids. Because of your generosity — this year, we are able to do just that.
From Lab Discovery to Clinical Care
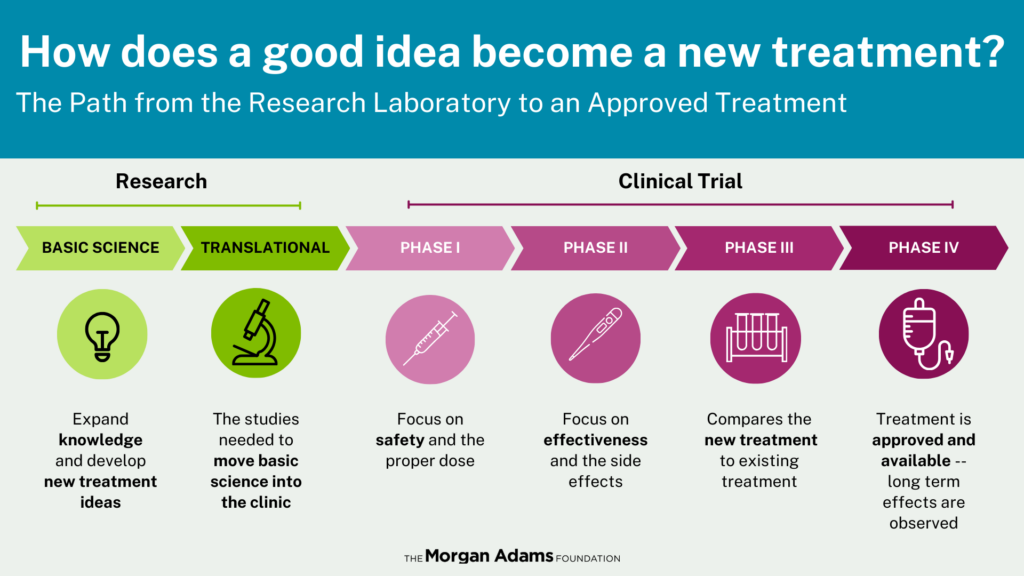
In pediatric oncology, clinical care and research are inseparable. Approximately 70% of children with cancer participate in some form of clinical trial. That is not because they are experimental subjects. It is because research is how pediatric cancer care has advanced and continues to advance.
Why Pediatric Trials are Different
Children are not just small adults. Their organs are still developing. Their brains, bones, and muscles are growing. A drug that is tolerated in an adult may behave very differently in a toddler or teenager.
That means pediatric oncologists cannot simply scale down an adult dose. They must carefully study how drugs move through and affect a child’s body and determine a dose that is both safe and biologically effective.
Because childhood cancers are more rare than adult cancers, collaboration is essential. Across the United States, researchers must work together through national consortia to execute them – because no single hospital sees enough patients to answer these questions alone.
Why Clinical Trials Matter
For children whose cancer has relapsed, or who never had effective options to begin with, early-phase trials are a lifeline – and the bridge between discovery and hope.
Next month, a longstanding national pediatric cancer clinical trial consortium will close, creating a critical gap in early-phase research infrastructure just as drug development momentum is accelerating.
The Morgan Adams Foundation is stepping in to help bridge that gap. Because of supporters like you, progress does not have to pause. Together, we are accelerating the future of pediatric cancer care!

What You and The Morgan Adams Foundation Are Doing to Help
In 2026, we are allocating funds for two Clinical Trials via national consortia to help push forward lab research previously funded by The Morgan Adams Foundation.
CONNECT is an international collaboration of world-class pediatric cancer centers, pharmaceutical companies, community partners and external consultants to facilitate and expedite research and clinical trials in high-risk brain tumors.
Dr. Foreman and his team at Children’s Hospital Colorado will collaborate with other COG institutions to further the preliminary studies of the combination two drugs that have demonstrated significantly improved outcomes for children with these tumors.
Want to learn more about the science behind this and other studies you’re helping to fund?
Learning more about the lives you’re supporting
Meet Kian
Kian was a typical rambunctious and playful 3-year-old boy, when he started complaining that his “head hurt” and began having difficulty with his balance. Upon the advice of a pediatric neurologist, he was sent to the ER where a CT scan revealed a substantial tumor.
From there, Kian was transported by ambulance to the Pediatric Intensive Care Unit at Children’s Hospital Colorado, where a team of doctors began care. Kian had his first ten-hour surgery three days later. The diagnosis was a Stage IV malignant Ependymoma. As a result of the surgery, Kian temporarily lost mobility on his right side, as well as speech and swallowing abilities.
Before he started his follow-up radiation treatment, his doctors discovered that the tumor had regrown. This finding resulted in two rounds of chemotherapy and a second 10-hour surgery just three months after his first. Fortunately, the second surgery resulted in no neurological damage, so he was able to promptly begin 33 treatments of radiation therapy, followed by two more rounds of chemotherapy.
Ependymoma tumors like Kian’s can often have poor outcomes when using standard therapies, especially for kids whose tumors recur. A clinical trial created by Dr. Nicholas Foreman and Dr. Katie Dorris combined two proven drugs, one that targets a highly expressed protein in ependymoma and another that triggers cancer cell death. Kian was able to join this trial – and thankfully had a good outcome.

Today, Kian is 16 years old and in the 10th grade. He enjoys science fiction, swimming, boating, building models of anything, watching movies, playing cards, and is learning to play chess. He has an extremely strong work ethic, which helps to counteract the deficits he has as a result of his cancer treatment. His biggest challenge is his processing speed, which causes delays in his ability to perform school-related tasks and in communication.
Cancer was an extremely destructive force on Kian (as it can be for so many kids), and it’s important for Kian’s parents to communicate that while his being cancer free is by far their biggest blessing, survivorship is far from perfect. It’s a testament to the need for safer, less-harmful treatments…and the profound importance of ongoing clinical trials.
To support groundbreaking kids’ cancer research like this click here.
Want to learn more about the science behind this and other studies you’re helping to fund?